
Theoretical cross-sections for Be and Li fragments differ strongly among the different models and from measured values. Oxygen is in group 6 of the periodic table, so it requires 6 valence electrons to balance out the nuclear charge. The reason the carbon atom needs a second orbital are complex and are beyond the. calculated with the effective nuclear charge. Step 3: determine whether valence electrons balance the nuclear charge out. In addition, there are now 6 electrons orbiting the nucleus in two orbitals. Carbon is the sixth element, with a ground-state electron configuration of 1s 2 2s 2 2p 2, of which the four outer electrons are valence electrons. 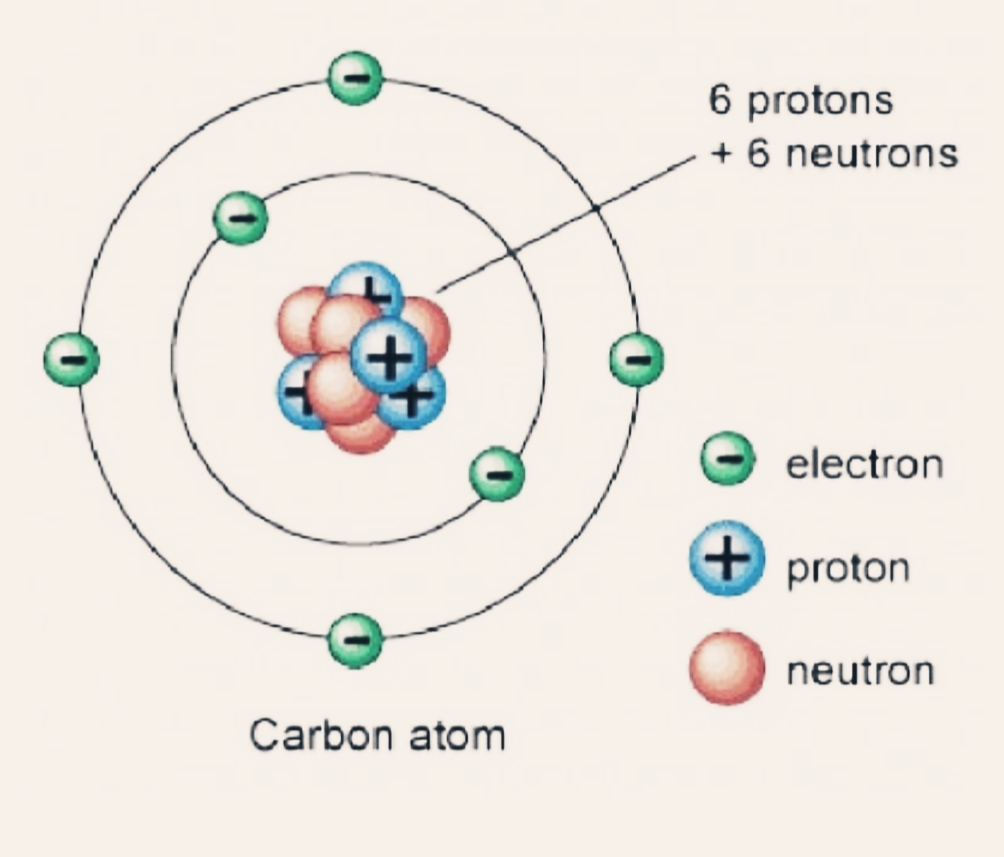
Each entry has a full citation identifying its source. (a) The picture of the displacement of the charges from the carbon nuclei along the normal of the aromatic plane. 
Two model calculations for production of B fragment are in good agreement with our measured data while a third model overestimates it by approximately 12%. Our carbon page has over 250 facts that span 117 different quantities. It has been found that the measured total charge removal cross-section agrees with theoretical predictions within approximately 10% and very well with previous experiments in corresponding energy regions. Description The effective atomic number Z eff, (sometimes referred to as the effective nuclear charge) of an atom is the number of protons that an electron in the element effectively 'sees' due to screening by inner-shell electrons. However, ESM does not add the extra charge to its prices but rather uses it as. 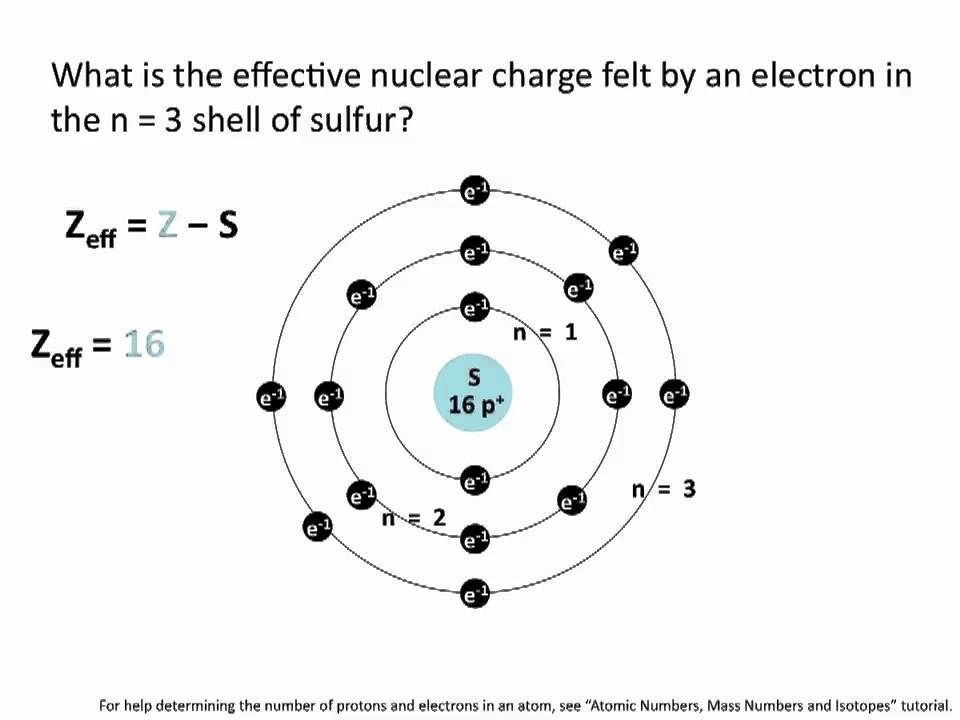
Also, it is noted that basis functions for the pseudopotential of the virtual. carbon pricing, phasing out coal and turning to renewables in. After analysis of the nuclear charge distributions, the total charge removal cross-sections and elemental production cross-sections of fragments with atomic numbers from 5 to 3, were obtained down to the lower energies (approximately 50 and 100 MeV/u, respectively). The above example is for a virtual atom on the way of carbon and nitrogen atoms. Valence electrons in C are closer to the nucleus (n 2) and are shielded only by the He core, so they experience greater attraction for the nucleus and have. Sources, facts, uses, scarcity (SRI), podcasts, alchemical symbols, videos and images. Track radius distributions representing the charge composition of the fragmented beams were automatically measured by a particle track analysis system. Element Carbon (C), Group 14, Atomic Number 6, p-block, Mass 12.011. \) experienced by the valence electrons in the magnesium anion, the neutral magnesium atom, and magnesium cation? Use the simple approximation for shielding constants.Stacks consisting of thin CR-39 sheets sandwiched between thick lucite and water absorbers were perpendicularly bombarded by 12C ions at 200 and 244 MeV/u.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
